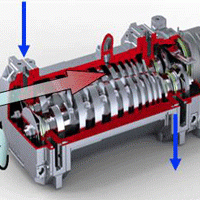
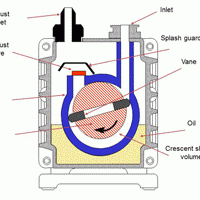
Oil sealed rotary vane pumps (aka rotary vane pumps) are the primary pumps on most vacuum systems used in the heat treatment industry. They are also referred to as a “backing” pump when used in combination with a booster pump, or with both a booster and secondary (“high vacuum”) pump, typically a diffusion style. A rotary vane pump can also be used alone when high vacuum is not required and slower pump-down is acceptable.
Two-stage designs are available, which utilize two rotors in series internal to the pump. Single-stage designs can provide a vacuum of 3 x 10-2 Torr (4 x 10-2 mbar), while two-stage designs can achieve 3 x 10-3 Torr (4 x 10-3 mbar). Due to the prevalence of rotary vane pumps, it is important for designers and users of industrial vacuum equipment to have a good understanding of how these pumps function. This series of articles will cover pump principles of operation, pump designs, pump oils, single-stage versus two-stage pump designs, contamination and gas ballast (manual and automatic), common accessories, applications, troubleshooting and pump maintenance.
Principles of Operation
Of the various vacuum pump technologies, rotary vane pumps are considered wet, positive displacement pumps. They are often called “wet” pumps because the gas being pumped is exposed to oil used as a lubricant to help provide the seal. For this reason, the oil is carefully selected and specially designed for the application. Positive displacement indicates that the pump works by mechanically trapping a volume of gas and moving it through the pump, creating a low pressure on the inlet side.