OAKVILLE, Ontario, (October 24, 2011) – First established in 2003 in Kalisz, Poland to offer Metallurgical Services to the burgeoning aerospace industry in Poland, VAC AERO has now opened a second facility located in the town of Rzeszow in southeast Poland. VAC AERO’s Polish divisions offer special processing services, including vacuum heat treating and brazing, vacuum carburizing, air plasma spray (APS) and HVOF protective coatings, as well as corrosion and oxidation resistant paint coatings. This new addition to the VAC AERO Group of Companies will be the sixth vacuum heat treating, brazing and coating facility worldwide.
Archives for December 2011
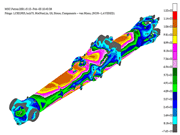
Landing-Gear Heat Treatment
The main components of a landing-gear structure are wheels and brakes, axles, bogie beams (a.k.a. truck beams), shock absorbers (a.k.a. shock struts), and drag and side braces. Primary design considerations on landing gear include maximum sink speed, spin up, spring back, lateral drift (on landing), towing, jacking, turning, braked roll, taxi, rebound, pivoting (main landing gear only), crashworthiness and fatigue.
Secondary loads include retraction/extension, aerodynamic loads, lock/unlock loads and emergency extension. In addition, nose landing-gear specific forces include dynamic breaking, nose-gear yaw and steering. Alloys used in these applications must have high strengths, normally between 260-300 ksi (1,792-2,068 MPa). By Carmine Filice, Daniel H. Herring, Paul Vanderpol
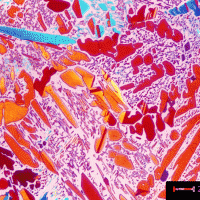
Microstructure of Nitrided Steels
Nitriding is one of the most interesting and useful surface-hardening techniques. It is unique in that during the nitriding process, the specimen is not heated into the austenite phase, and it does not rely upon the formation of martensite to achieve high hardness and useful properties. It is heat treated prior to nitriding, forming tempered martensite to obtain the desired core properties unlike all other surface heat-treatment processes.
The processing associated with nitriding does have some advantages in avoiding problems such as quench cracking and distortion. It also has some side benefits in improved corrosion resistance and generation of beneficial residual compressive stresses, which improves fatigue resistance. Nitrided surfaces do exhibit high surface hardness, leading to improved wear resistance.
Introduction to Stereological Principles
Although the fundamental relationships for stereology, the foundation of quantitative metallography, have been known for some time, implementation of these concepts has been restricted when performed manually due to the tremendous effort required. Further, while humans are quite good with pattern recognition, as in the identification of complex structures, they are less satisfactory for repetitive counting.
Many years ago, George Moore (1) and members of ASTM Committee E-4 on Metallography conducted a simple counting experiment. About 400 people were asked to count the number of times the letter “e” appeared in a paragraph without striking out the letters as they counted. The correct answer was obtained by only 3.8% of the people. Results were not Gaussian, however, as only 4.3% had higher values while 92% had lower values, some much lower. The standard deviation was 12.28. This experiment revealed a basic problem with manual ratings. In this case the subject was one very familiar to the test subjects, yet only 3.8% obtained the correct count. What degree of counting accuracy can be expected when the subject is less familiar, such as microstructural features? Image analyzers, on the other hand, are quite good at counting but not as competent at recognizing features of interest. Fortunately, there has been tremendous progress in the development of powerful, user-friendly image analyzers over the past two decades.
Introduction to Quantitative Metallography
For most of its history, metallographic observations have been largely qualitative in nature. The structure might be described as being relatively coarse or fine, or layered, or uniform. Particles might be labeled as globular or spheroidal, lamellar, acicular, or blocky. Microstructures were single-phase or duplex, and so forth.
Forty some years ago when I entered industry, chart ratings and visual examinations were the main approach toward quantitation. I can well remember the mill metallographers looking at spheroidized carbide tool steel structures and stating that it was, for example, 90% spheroidized (many raters would never say 100%, just as some teachers would never grade an essay at 100%!) or that it was 60% spheroidized and 40% lamellar tending to spheroidize. Or, without looking at the chart (a seasoned rater never did!), they would pronounce that the grain size was, for example, 100% 6 to 8 or perhaps 70% 8 and 30% 3 to 5 if it was duplex in appearance.
Study of Selective Etching of Carbides in Steel
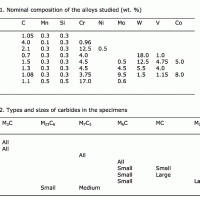
A number of etchants have been reported in the literature to selectively outline, outline and color, or attack specific types of carbides in steels. These etchants have been developed during the first half of the 20th century but have not been studied systematically since the development of modern analytical techniques. To evaluate these etchants, eight specimens (seven different compositions) with M3C, M23C6, M7C3, M6C, M2C and MC carbides were studied, first by electron-backscattered diffraction (EBSD) to verify the carbides present.
The matrix of each specimen was darkened to measure the total carbide content. Then, the various etchants were tried and the results were compared to past publication results. Quantitative measurements were made after each etchant was used. This revealed some minor differences with the prior literature and showed that, while useful for qualitative evaluations, they are not useful for quantitative measurements. Metallographers have used certain selective etchants for years to color specific carbides as an aid to phase identification. However, only a few of these etchants have had wide usage (e.g., alkaline sodium picrate to color cementite since 1906 due to the work by Kourbatoff). The behaviors of the others are less well known. Even for alkaline sodium picrate, however, few metallographers know that it also outlines and colors M6C carbides.